In the News
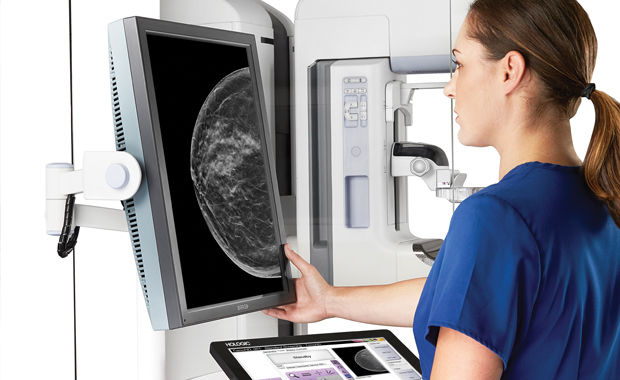
CureMetrix gets FDA clearance for mammogram-screening system
Source: San Diego Union-Tribune
La Jolla’s CureMetrix said Tuesday it has received FDA approval to sell its software “triage” service to screen mammograms for signs of cancer.
CureMetrix’s cmTriage flags suspect mammograms so radiologists can give them a closer look, said Kevin Harris, CEO of the privately held company.
The automated process enables radiologists to devote more of their attention to abnormal mammograms instead of having to look at every image to find them. The cloud-based service sends back the results in three to four minutes, Harris said.
“What we’re seeing in preliminary studies is the triage software can help doctors read through their work list up to 40 percent faster, which as you might expect, is a big impact on the clinical efficiency,” Harris said.
Patient identifying information is removed at the hospital by the service, which is designed to work with all mammography systems, he said.


