COVID-19 News
COVID-19 Update | February 17, 2023
February 17, 2023

COVID-19 Update | February 17, 2023
- Pfizer’s antiviral COVID-19 treatment, Paxlovid, is shown to elicit a strong immune response in older adults, significantly reducing the risk of hospitalizations and deaths from COVID-19.
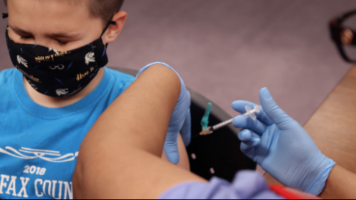
- A new study finds that a three-dose primary series of the Pfizer-BioNTech COVID-19 vaccine was found to be safe, immunogenic, and efficacious in young children.
- This Black History Month, we recognize the pivotal role Kizzmekia Corbett played in the development of the mRNA COVID-19 vaccine.
Recent News
- Paxlovid works well against COVID-19 in older adults, but not younger people: study
Fierce Healthcare – February 16, 2023
Paxlovid significantly reduces the risk of hospitalizations and death from COVID-19 in older individuals, but not for younger people, something payers and providers might want to keep in mind as the medication will stop being offered for free in the U.S. when the public health emergency ends in May. Researchers with Public Health Ontario stated in their study in the Canadian Medical Association Journal that “we observed substantial variation in the absolute risk reduction, which suggests that use of nirmatrelvir–ritonavir [Paxlovid] in populations at lower risk of COVID-19 may have limited population health benefits with important implications for its cost-effectiveness evaluations.” - Regulatory T Cells May Influence Severity of COVID-19 Among Sexes
Pharmacy Times – February 16, 2023
Circulating follicular regulatory T cells—a subset of regulatory T cells—and other Treg subsets may explain the link between sex and severity of COVID-19, according to a recent study published in PNAS. Tregs were found to proliferate in the T cells of critically ill patients, according to the investigators. Current understanding of the differences in antibody production between the male and female sexes during COVID-19 is murky, according to the investigators. Previous research associated the female sex with a greater antibody response and susceptibility to autoimmune diseases, but antibody levels and disease susceptibility in COVID-19 appears to be higher for males, according to the study. The effects are Tfr control plasma cell formation, antibody quality, B cell memory, and they protect the lungs against influenza. Tfr and Tregs may be important in understanding a person’s susceptibility and recovery from COVID-19. - Nirmatrelvir–ritonavir found to lower COVID-19 severity outcomes
News Medical Life Science – February 16, 2023
In a recent study published in the Canadian Medical Association Journal, researchers evaluated the effectiveness of nirmatrelvir–ritonavir in preventing coronavirus disease 2019 (COVID-19) severity outcomes during the severe acute respiratory syndrome COVID-19 2 (SARS-CoV-2) Omicron variant predominance. Overall, the study findings showed that nirmatrelvir–ritonavir usage significantly reduced the chances of COVID-19-associated hospitalizations and deaths, underpinning nirmatrelvir–ritonavir use for mild COVID-19 patients at an elevated risk of severe illness. The greatest benefit was observed among under-vaccinated individuals and those aged ≥ 70.0 years. - Three Doses of Pfizer-BioNTech COVID-19 Vaccine is Efficacious in Young Children
Contagion Live – February 15, 2023
A three-dose primary series of (Pfizer-BioNTech) COVID-19 vaccine was found to be safe, immunogenic, and efficacious in children 6 months to 4 years of age, according to a new study. The study’s results were published in The New England Journal of Medicine. Overall, a vaccine efficacy of 73.2% was observed in individuals 6 months to 4 years of age. In children ages 6 months to less than 2 years of age, the vaccine demonstrated a 75.8% efficacy, and among children 2 to 4 years of age it was 71.8% efficacious. - Paxlovid Doesn’t Increase Risk of Rebound COVID-19 Infection: Study
WebMD – February 15, 2023
People who took the antiviral Paxlovid to treat COVID-19 infections were not more likely to get back-to-back bouts of the virus, a new study shows. The findings offer clarity amid concerns that the use of Paxlovid, which works by stopping the spread of the virus in the body, increased the risk of COVID-19 rebound. “Rebound is a re-emergence of symptoms and an uptick in viral load after a period of recovery,” the Center for Infectious Disease Research and Policy explained in a summary of the study. - Reassuring Findings on Bivalent COVID-19 Booster in Hemodialysis Patients
MedPage Today – February 15, 2023
Anti-spike IgG concentrations against BA.4/5 significantly increased in patients with end-stage renal disease (ESRD) following a bivalent booster, particularly in those without a prior Omicron infection, a case series found. The study looked at 55 ESRD patients on hemodialysis who had received four prior doses of a monovalent vaccine, including about a third with prior Omicron breakthrough infections. From 2 weeks before to 2 weeks after a fifth dose with a bivalent mRNA vaccine, anti-spike IgG concentrations increased by a factor of 2.5 for patients with a prior infection and by a factor of 7.3 for those without a previous infection. - In about-face, Moderna vows Americans won’t have to pay for its COVID-19 vaccine
Stat News – February 15, 2023
In an unexpected shift, Moderna has decided not to ask Americans to pay for its COVID-19 vaccine, a move that follows intense criticism over initial plans to charge $110 to $130 per dose after the company pivots from government contracts to commercial distribution. The vaccine maker released a brief statement that it “remains committed” to ensuring everyone in the U.S. has access to its COVID-19 shot, regardless of whether they have health insurance coverage. For those lacking sufficient insurance, the company will tap a patient assistance program. “Everyone in the U.S. will have access to Moderna’s COVID-19 vaccine regardless of their ability to pay,” the company said. - CDC updates vaccination schedule for children and adolescents, includes COVID-19 vaccine
Contemporary Pediatrics – February 14, 2023
The CDC’s Advisory Committee on Immunization Practices (ACIP) announced the updated 2023 immunization schedule for children and adolescents under 18 years of age. The published new recommendations for families and clinicians are part of routine vaccinations as well as inclusion of the COVID-19 vaccine. According to the CDC, the following are the COVID-19 vaccine recommendations for the primary series: Age 6 months–4 years: 2-dose series at 0, 4-8 weeks (Moderna) or 3-dose series at 0, 3-8, 11-16 weeks (Pfizer-BioNTech). Age 5–11 years: 2-dose series at 0, 4-8 weeks (Moderna) or 2-dose series at 0, 3-8 weeks (Pfizer-BioNTech). Age 12–18 years: 2-dose series at 0, 4-8 weeks (Moderna) or 2-dose series at 0, 3-8 weeks (Novavax, Pfizer-BioNTech) - She Helped Unlock the Science of the COVID-19 Vaccine
The New York Times – February 13, 2023
Kizzmekia Corbett had gone home to North Carolina for the holidays in 2019 when the headlines began to trickle in: A strange, pneumonialike illness was making dozens of people sick in China. By the first week of January 2020, the number of infected people in China had climbed to the hundreds, and Dr. Corbett, a viral immunologist, was back at her desk at the National Institutes of Health, where she served as a senior research fellow at the Vaccine Research Center at the National Institute of Allergy and Infectious Diseases. And that’s when the news was confirmed: The mysterious illness was a novel COVID-19, exactly the category of infection that she had been probing for the past five years in a bid to develop a vaccine.
Rely on California Biotechnology Foundation to monitor breaking news and provide updates on the latest advancements in COVID-19 diagnostics, vaccines and treatments.
Stay informed on the latest news and trends on the economic and health benefits of this industry by visiting the new CABiotech.org
If you have any questions about informational briefings contact California Biotechnology Foundation Executive Director Patty Cooper at (916)764-2434 or [email protected].